Wolfgang Baehr

Professor of Ophthalmology & Visual Sciences
Adjunct Professor of Neurobiology
Adjunct Professor of School of Biological Sciences
Neurobiology of Disease
Molecular Neurobiology
Email:
Education:
Ph.D. 1970, University of Heidelberg, Germany; Postdoctoral Fellow, 1971-1975, Max-Planck Institute of Biophysical Chemistry, Goettingen, Germany
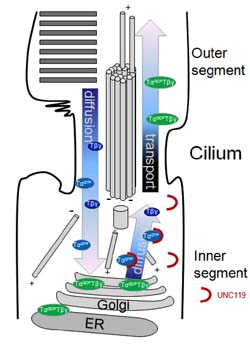
Model of light induced transpocation of transducin in rod photoreceptors. In light,
as part of a desensitization mechanism, transducin migrates to the inner segment.
In dark, it slowly returns. Mechanisms of translocation are controversial, and tought
to be based on diffusion, vesicular transport, or a combinantion of both.
RESEARCH:
Phototransduction, the retinoid cycle, membrane protein transport in photoreceptors and mouse models for inherited retinal disease
Rod and cone photoreceptors have evolved into highly polarized structures consisting of three distinct areas: the outer segment containing membrane disks housing proteins involved in phototransduction, the inner segment where biosynthesis occurs, and the synaptic terminal that transmits excitation by light to downstream neurons. The inner segment (cell body) connects to an outer segment through a narrow 9+0 cilium, and to the synaptic terminal by a slender axon. Outer segments of rods and cones are renewed roughly every ten days. New disks are made at the proximal end, old disks are shed at the distal end, and phagocytosed by the adjacent retinal pigment epithelium (RPE). Daily renewal of ~10% (about 100 disks) of the outer segment membrane requires a high rate of biosynthesis to replace OS proteins, with reliable transport and targeting pathways.
My laboratory explores mechanisms in membrane protein transport in mammalian rod and cone photoreceptors, specifically post-biosynthesis transport of integral membrane and peripheral membrane-associated proteins to the outer segments. Integral membrane proteins are synthesized at ER-associated ribosomes and exported to the Golgi apparatus. Peripheral membrane proteins are synthesized in the cytosol and become ER-associated if prenylated or acylated. Vesicles emerge from the trans-Golgi network (TGN) and transport to the base of the cilium where they fuse with the cell membrane. Finally, cargo is assembled for intraflagellar transport to the outer segment where phototransduction occurs.
We are interested in proteins/genes mediating transport, particularly molecular motors (kinesin-II), small GTP binding proteins (rab8, rab11), prenyl binding proteins mediating transport of prenylated proteins (PrBP/delta or PDE6D), acyl binding proteins (UNC119) involved in transport of G protein subunits, and centrins, small Ca2+ binding proteins involved in ciliogenesis. We produce knockouts/knockins, transgenics to monitor the consequences of gene knockouts, dominant negative transgenes, or short hairpin RNAi. Most frequently applied techniques are molecular biology, confocal and electron microscopy, optomotry (behaviour), in-vivo electroporation, and gene therapy (AAV virus).
