Oleksandr (Alex) Shcheglovitov

Assistant Professor of Neurobiology
Shcheglovitov lab
Cellular Neuroscience
Molecular Neuroscience
Developmental Neurobiology
Neurobiology of Disease
E-mail:
Education
B.S. 2000, National Technical University of Ukraine "KPI", Kyiv, Ukraine; M.S. 2002, National Technical University of Ukraine "KPI", Kyiv, Ukraine; Ph.D. 2007, Bogomoletz Institute of Physiology, Kyiv, Ukraine; Postdoctoral Fellow 2007-2009, University of Virginia; Postdoctoral Fellow 2009-2014, Stanford University
RESEARCH:
Development and function of human synapses in health and disease
Synapses provide the major biochemical substrates for the development and execution of cognitive functions in the brain. Correspondingly, mutations in synaptic proteins have been identified in patients with various mental illnesses. Due to the very limited access to the human brain, mechanisms of synaptic development and function have been mainly investigated in animal models. However, major differences between animals and humans make it difficult to translate the results acquired in animals into treatments for patients. As a result, medicines that are currently available for patients with autism, intellectual disability, bipolar disorders, depression, schizophrenia, and other neuropsychiatric disorders are largely unspecific with serious side effects. Therefore, there is a critical need to study synapses in human neurons. We study the development and function of human synapses under normal and pathological conditions using human neurons derived from induced pluripotent stem cells (iPSCs).
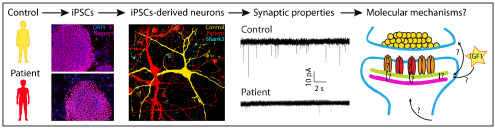
We start with skin cells of patients with different neuropsychiatric disorders that carry specific genetic abnormalities in genes encoding synaptic proteins. We reprogram skin cells into iPSCs and differentiate iPSCs into neurons. We study synaptic properties of human neurons using electrophysiology, imaging, biochemistry, and molecular biology techniques. We seek to understand the molecular mechanisms underlying the development and function of human synapses under normal and pathological conditions.
My Bibliography:
Google Scholar: http://scholar.google.com/citations?user=99Y5cegAAAAJ&hl=en
